The energy levels of solids and solutions reverse in order of height. Calculate the lattice enthalpy of KCl from the following data at standard statesEnthalpy of sublimation of K 89 kJ mol 1 Enthalpy of dissociation of Cl. And we can say here, as we increase the lattice energy are ionic bond gets stronger, so the ions air helped more tightly together. Here, we can say recall an increase in a lattice energy causes an increase in the strength of an ionic bond. The enthalpies of solution for some salts can be positive values, in these cases the temperatures of the solution decrease as the substances dissolve the dissolving is an endothermic reaction. The lattice energies of KF,KCl,KBr and KI follows the order: A KF>KCl>KBr>KI B KI>KBr>KCL>KF C KF>KCl>KI>Br D KI>KBr>KF>KCl Medium Solution Verified by Toppr Correct option is A) Lattice energy is directly proportional to the charge of the ions and inversely proportional to the size of the ions. Now the lattice energy of an ionic solid has a direct impact on its physical properties. That means that we will have to use theoretical values of their lattice enthalpies.\) refer to the same quantity, and they are used interchangably. In order to continue to use our website, we ask you to confirm your identity as a person. Let's look at this in terms of Born-Haber cycles of and contrast the enthalpy change of formation for the imaginary compounds MgCl and MgCl 3. It turns out that MgCl 2 is the formula of the compound which has the most negative enthalpy change of formation - in other words, it is the most stable one relative to the elements magnesium and chlorine. kJ/mol Ionization energy for K 419 kJ/mol Electron affinity of Cl 349 kJ/mol Bond energy of Cl2 239 kJ/mol Energy of sublimation for K 90. 
Generally, lattice energy is inversely proportional to the size of the ions and. Lattice enthalpy of NaCl is 787 KJ/mol Lattice enthalpy of NaOH is 898 KJ/ mol. Use the following data for potassium chloride to estimate E for the reaction: K(s)+12Cl2(g)KCL(s)E Lattice energy 690. Crystals cleave easily in three directions.  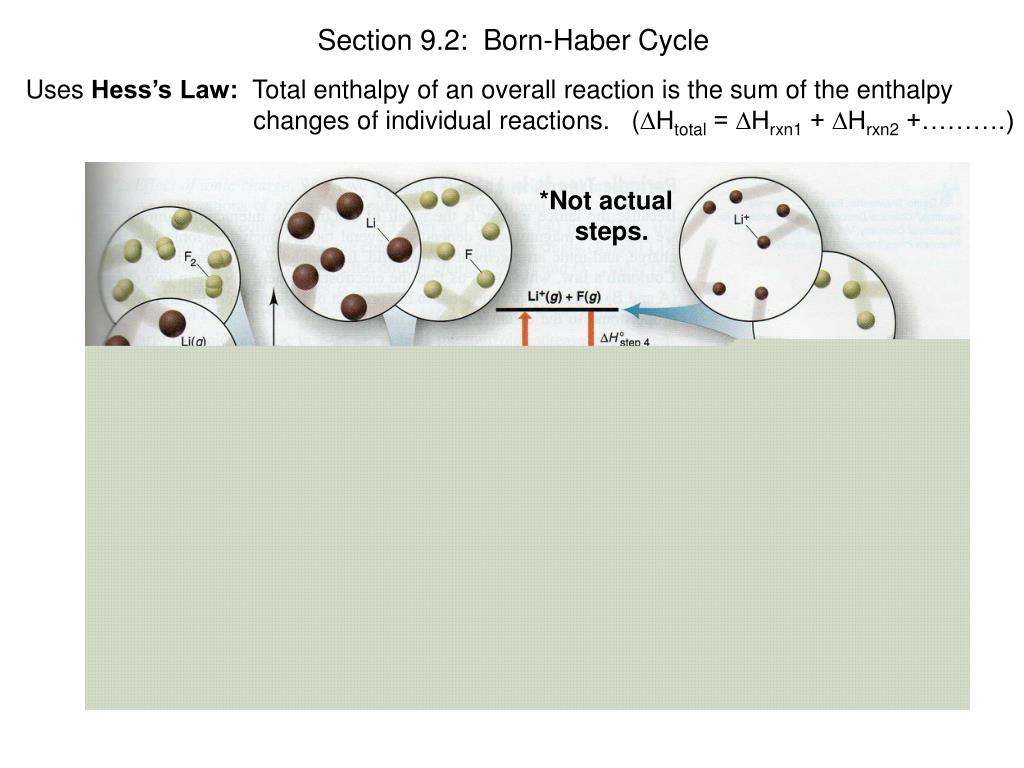
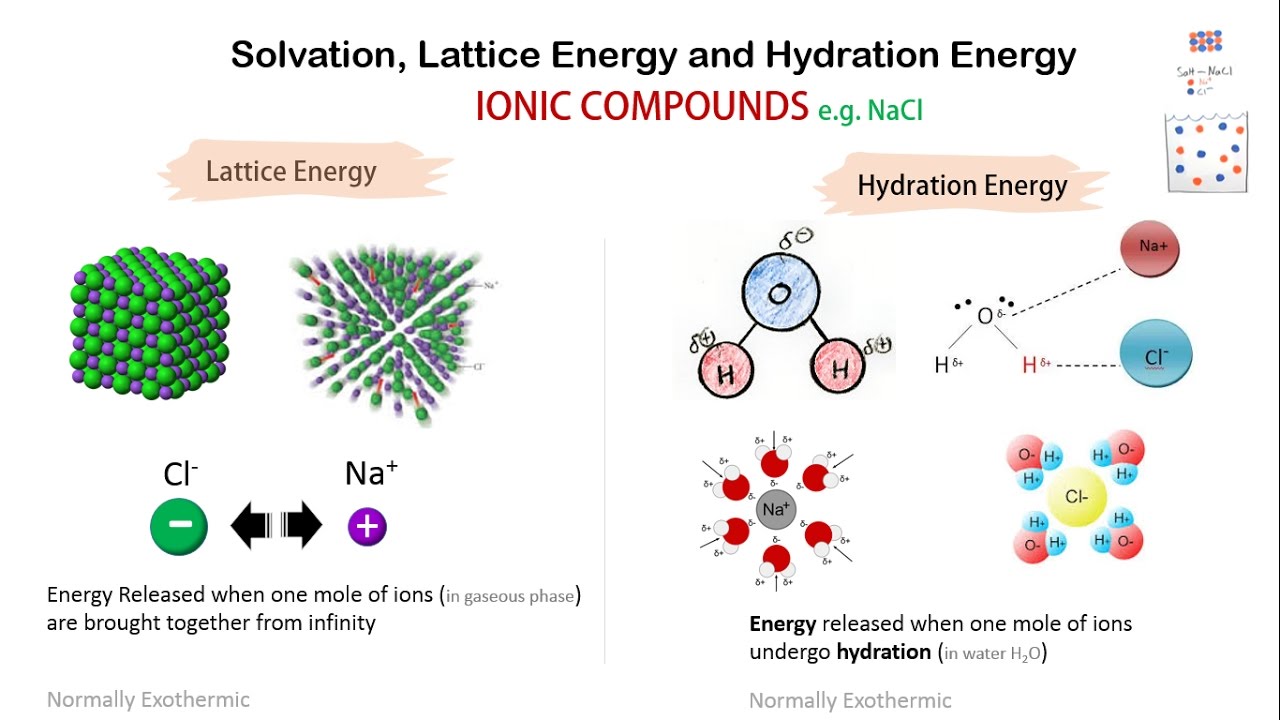
Hence, it is typically used to measure the bond strength of ionic compounds. It adopts a face-centered cubic structure known as the B1 phase with a lattice constant of roughly 6.3. 3,4 Ionic salts have gained substantial importa. Lattice energy can be defined as a measure of the energy required to dissociate one (1) mole of an ionic compound into its constituent anions and cations, in the gaseous state. 1,2 Ionic salts like NaCl and KCl are essential to maintain osmotic pressure inside the cell as well in signal transmission across the cells. The question arises as to why, from an energetics point of view, magnesium chloride is MgCl 2 rather than MgCl or MgCl 3 (or any other formula you might like to choose). Ionic compounds are ubiquitous materials and are characterized by their highly crystalline nature, high melting points and strong miscibility in polar media. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
